 
This article was written for you by Samantha, one of the tutors with TestPrep Academy. As you can see that the distance between two adjacent Hydrogen atoms is measured and then its half value will give you the atomic. Above image clearly shows you how the atomic radius is measured. Looking to get ready for the ACT? We can help with ACT Prep Atomic radius is measured by measuring the distance between the two adjacent atoms, and then dividing that distance by 2, we get the atomic radius. Atomic radius is the distance from the atom’s nucleus to the outer edge of the electron cloud. It is important to note that elements in the same period all have the same number of electrons shells, so electron shielding will not be a factor.Ītomic size increases as you go down a column because of the addition of another electron shell and electron shielding.Ītomic size decreases as you go right across a row because of increased protons. Atomic radius increases Atomic radius increases. Thus, the electrons are held more tightly towards the nucleus, decreasing the radius. As you move across a period Why Decrease because of the increasing positive charge on the electrons exerted by the increasing number of protons. The increase in positive charge increases the attraction between the nucleus and the electrons of the atom. As the number of protons increase, the nucleus of the atom becomes more positively charged. Electrons are added to the same principal. This is because the number of protons increase moving to the right of the row. Across Period 3, proton number increases, the nucleus is more positively charged hence nuclear charge increases. When moving left to right across a period, the atomic size decreases. Going Left and Right Across Periods (Rows) 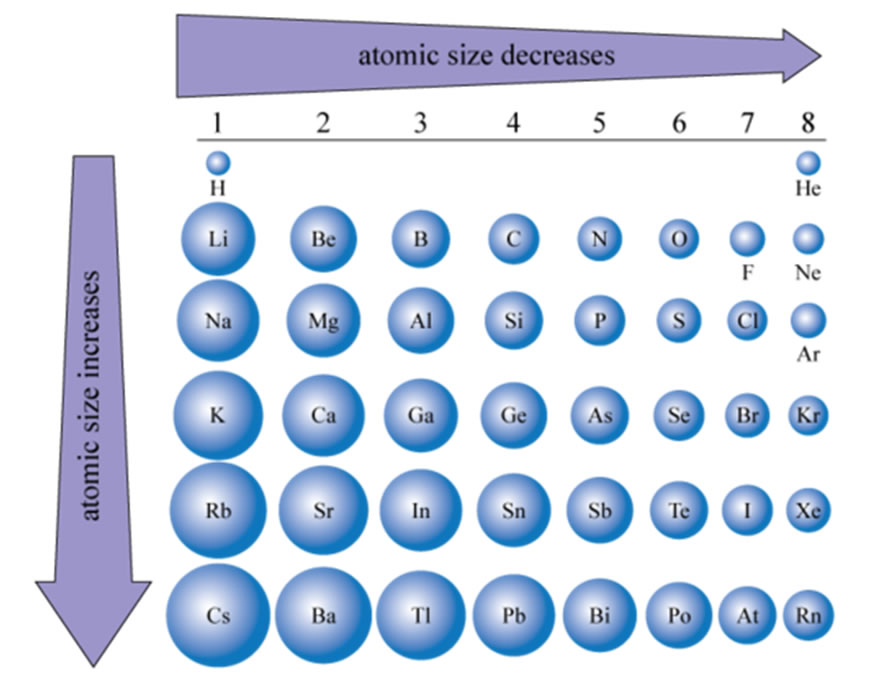
As a result, the electrons are not held as tightly towards the nucleus. In addition, the electron shells in between the valence shell and nucleus present electron shielding that also minimizes the attraction. Also, the new orbital is further away from the nucleus, meaning the attraction between the positively charged nucleus and the valence electrons decrease. Electronegativity decreases across a period and decreases down a group. This is because as you go down the period table, new valence shells are added and thus, increasing the radius. Atomic radius decreases across a period and increases down a group. When moving down a group, the atomic size increases. The atomic size, or atomic radius, is the distance between the nucleus of an atom to the outermost electron orbital, where the valence electrons are. 
These patterns are called periodic trends. The tendencies of these patterns increase or decrease as you move along the columns and rows of the table. Explain the large decrease in atomic radii as you move across a period from. These electrons screen or shield the outer electrons from the nuclear charge.Specific patterns of certain elemental characteristics are present in the periodic table. The nuclear charge of the atom increases across a period because there are more protons concentrated in the nucleus of the atom. The amount of energy needed to remove an electron from a specific atom is. This is because of the screening effect of the filled inner electron levels. The greater attraction between the increased number of protons (increased nuclear charge) and electrons, pulls the electrons closer together, hence the smaller size.Īs you move down a group in the periodic table, the covalent radius increases. These electrons are relatively ineffective at. The covalent radius (a measure of how large individual atoms are) shows different trends if you are moving across a period or down a group.Ī comparison of the relative covalent radii of atoms is shown in the diagram below.Īcross a period from left to right, the covalent radius decreases.Īs you move from left to right across the periodic table, atoms have more electrons in their outer energy level and more protons in their nucleus. As you go across the third period from Na to Ar, each additional electron adds to the same shell (n 3). Periodic trends, arising from the arrangement of the periodic table, provide chemists with an invaluable tool to quickly predict an element's properties. The atomic radius moving down an element period or column tends to increase because an additional electron shell is added for each new row. Patterns and trends in the periodic tableĬhemists observe patterns in different properties of elements as they are arranged in the periodic table. Thats because the electrons become more tightly packed as you move across the periodic table, so while there are more electrons for elements of increasing atomic number, the atomic radius may decrease. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
